
Single-Session1*
*Complete tumour regression in 98.5% of lesions treated, with 89% after a single application.3
Non-invasive†
†A procedure is considered non-invasive when no break or cut in the skin is created.6
Aesthetic / functional1,2§
§No disfiguring scarring; Retained functionality in high-risk and difficult to treat areas.
Short duration of treatment1,2¶
¶Treatment time is generally 30 to 180 minutes.
Precise1,3,4**
**Accurate dosing. Spares healthy tissue.
Painless1,4‡
‡No reported pain.1,4
Patient identification
- Single or multiple lesions
- Patients with comorbidities
- Lesion is difficult to treat
- Lesion is hard to reach
Indications
- Basal call carcinomas (BCC)
- Squamous cell carcinomas (SCC)
- Erythroplasia of Queyrat
- Bowen’s disease
Watch these video to understand more about the treatment
This is a short demonstration video of the Rhenium-SCT, showing the application device and treatment process.
This is a documentary video discussing the burden of disease, treatment considerations and Rhenium-SCT.
Rhenium-SCT®: Application System
Engineered to ensure safety, convenience and quality control.
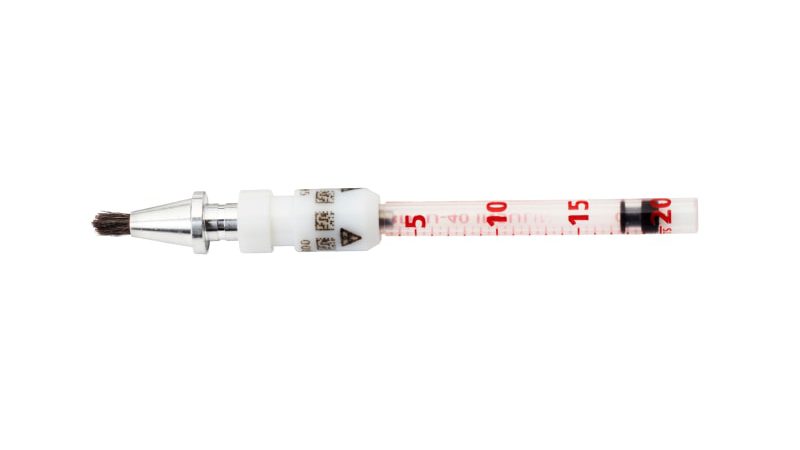
Carpoule
Contains Renium-188 in the form of a paste:
- Max. activity per unit: 10 GBq.
- Max. activity at admin: 2.2 GBq.

Applicator
For safe, ergonomic, handling of carpoules:
- Rhenium-188 carpoule is loaded into the applicator.
- Allows a targeted application of the compound to the special foil above the lesion.
- Offers the user protection and precise handling.

Base Station
Transport Unit, containing the carpoules, is inserted into the base station:
- Homogenises the radioactive compound inside the carpoule.
- Loads the carpoule into the applicator.

Measurement Station
Determines the activity of the carpoule before and after each application:
- The applicator is positioned in the Measurement Station to perform the activity measurements.
- In order to calculate the treatment time, the Carpoule is again measured for residual activity after the application.
Precise, personalised treatment
Rhenium-SCT® (Skin Cancer Therapy) is a brachytherapy utilising the Beta emitter radioisotope Rhenium-188 for the treatment Non-Melanoma Skin Cancer.
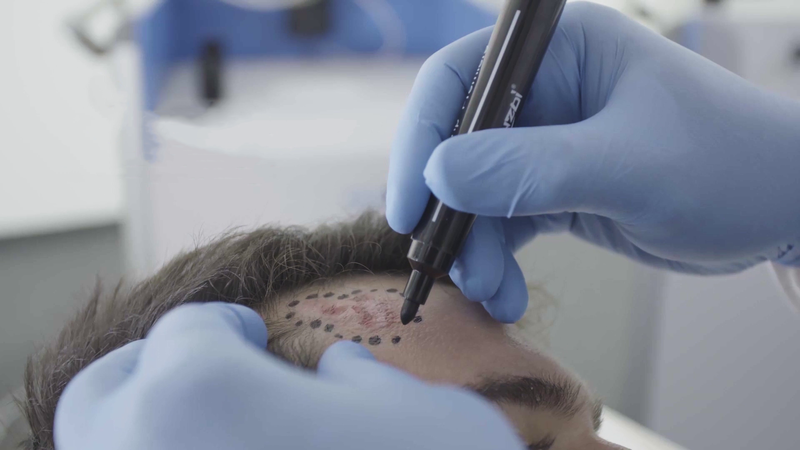
Step one
The physician marks the area to be treated.

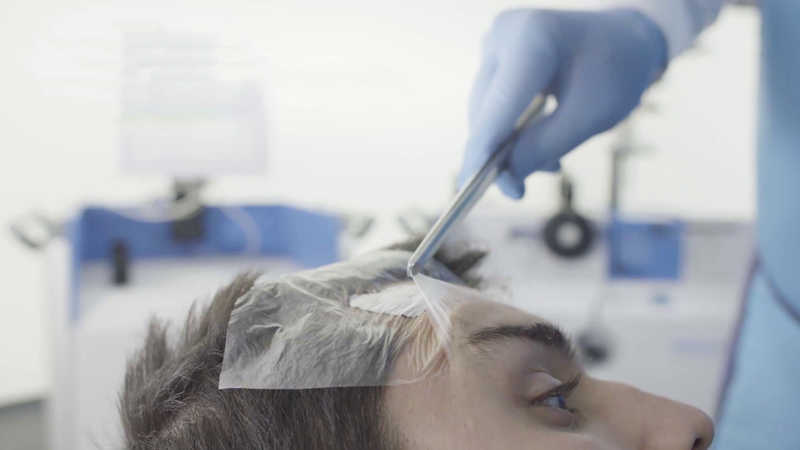
Step two
The area is covered with a clear film before applying the Rhenium-SCT® paste.
Step three
After a calculated period of time (between 30-180mins), the film is removed and the waste is disposed of.

Available resources for your clinic
We have prepared materials for use in clinic waiting rooms, treatment consideration tools and patient information.

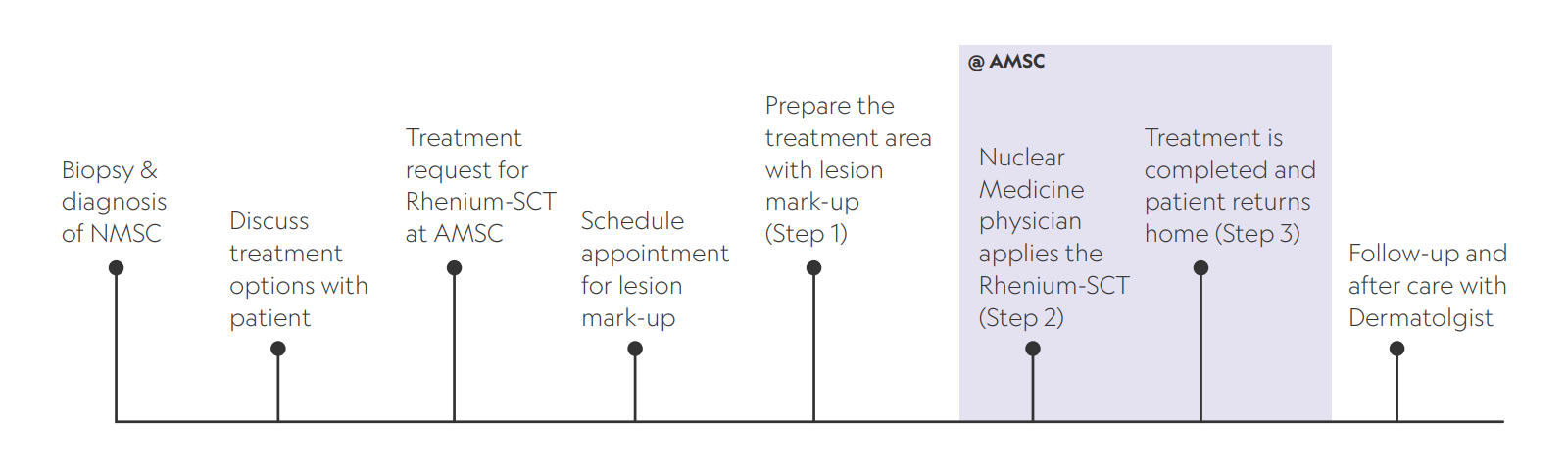
Understanding the patient pathway
Your patients remain in your care throughout the process – AMSC are simply the providers of this specialist treatment with a licensed nuclear medicine physician.

Developed in Germany. Researched Globally. Now Available in Melbourne.
Avion Medical Skin Centres (AMSC) have been working with OncoBeta GmbH in bringing this innovative therapy to Australia. Rhenium-SCT® (skin cancer therapy) is now available via our state-of-the-art treatment facility within the Melbourne Theranostics Innovation Centre (MTIC). We are pleased to offer this alternative to traditional invasive options, designed to provide improved cosmetic outcomes, in a painless, single-session treatment.1,4*†
*Complete tumour regression in 98.5% of lesions treated3
†No reported pain1,4
Treatment Requests & Enquiries
Physicians can use this form to submit their patients’ online Treatment Request Form, or contact us for more information, request a rep follow-up and materials to use in clinics. Physicians, Patients and everyone else can use this form for all General Enquiries too.
Appointment Request for Patients
Treatment Request for HCPs
General Enquiries
Disclaimer
This information is intended for use by Healthcare Professionals in Australia, only. By clicking “continue” you acknowledge that you are an Australian Healthcare Professional.
